The LLNL SiC Mystery
I’ve posted previously about some oddities in the LLNL Light Armor Program’s ceramic test results. There’s another oddity that, in itself, was relatively minor, but proved unfortunately influential for decades: Their experiments with silicon carbide.
As you can see in the table below, silicon carbide (SiC) didn’t perform very well. Its figure of merit was 0.76 – well under boron carbide’s (B4C) baseline of 1.0, and only marginally better than high-purity alumina at 0.73. In practical terms: if it would take a 10-pound B4C tile to offer a given level of protection, you’d need a 13.16-pound SiC tile, or a 13.70-pound alumina tile.
That’s on a weight basis. On a thickness basis, it gets even worse: the LLNL’s SiC was the worst-performing material of the three. Their SiC required approximately 7% more thickness than B4C and 24% more thickness than alumina to provide equivalent ballistic protection.
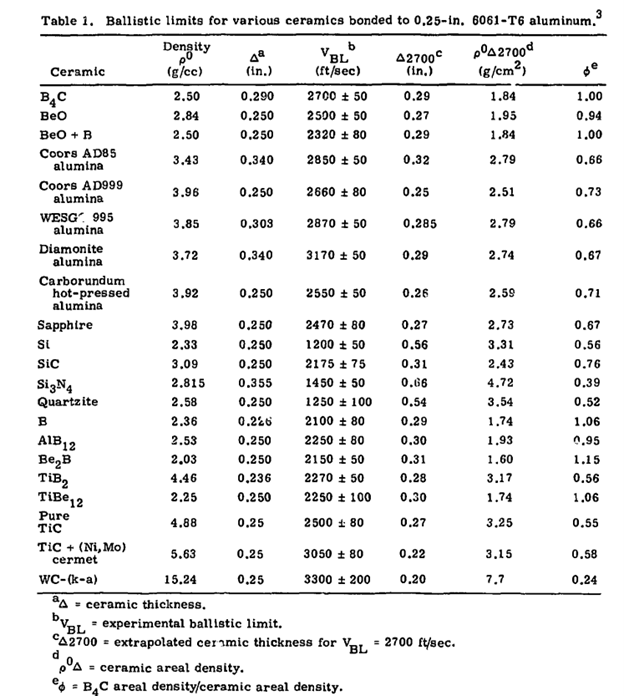
Yet today SiC totally reverses this paradigm, outperforming both B4C and Al2O3 on a thickness basis. In modern ceramic armor plate production, given a certain margin of safety, a 7.8mm thickness of high-quality SiC will reliably stop a common reference AP threat, whereas high-grade boron carbide requires no less than 8.3mm; in this case, SiC needs approximately 6% less thickness than B4C. Compared against alumina, the relation varies (alumina’s performance depends heavily on its compressive strength, hardness, and density, all of which track with purity grade), but SiC generally outperforms Al2O3 on both thickness and weight.
In fact, the best grades of modern SiC approach the weight efficacy of today’s best grades of boron carbide, and militaries around the world have been moving to SiC as their workhorse ceramic armor material.
So why did the LLNL report such poor results?
The answer is that their tests were run in the late 1960s, and before 1974 high-quality SiC was effectively unsinterable. They were testing “SiC” that by today’s standards would be regarded as unfit for purpose.
The Sintering Problem
In the 1960s, the only known way to densify non-oxide ceramics such as carbides and borides to their theoretical full density was via hot-pressing. B4C and most other non-oxide ceramics were highly amenable to this process – but SiC was stubbornly resistant. It proved almost impossible to produce high-quality SiC via the straightforward hot-pressing of pure SiC powders.
The problem is thermodynamic. At elevated temperatures, silicon carbide doesn’t simply soften and flow like most ceramics. Instead, it decomposes: the silicon sublimates, leaving behind a porous carbon skeleton. This decomposition begins at temperatures well below those needed to achieve full densification through solid-state diffusion. Hot-pressing pure SiC powders typically yields a weak, porous body riddled with voids – or, if pushed too hard, a pile of graphite and silicon vapor.
Throughout the 1960s, researchers tried various workarounds. Some used extremely high pressures. Others experimented with additives. The results were inconsistent at best. The SiC that the LLNL tested was almost certainly produced by one of these early, imperfect methods – likely a hot-pressed material with significant residual porosity, decomposition damage, or both.
The Breakthrough
Everything changed in 1974, when Svante Prochazka at General Electric discovered that small additions of boron and carbon could enable pressureless sintering of SiC to near-theoretical density. The mechanism was elegant: Boron segregates to the grain boundaries and dramatically accelerates diffusion, while carbon removes the thin oxide layer (SiO2) that normally coats SiC particles and inhibits sintering.
This was a genuine revolution. Suddenly, high-density SiC could be produced in conventional furnaces without the massive hydraulic presses that hot-pressing required. Costs dropped. Quality improved. Complex shapes became feasible.
The sintered SiC that emerged from this process bore little resemblance to the porous, compromised material of the 1960s. It was fully dense, fine-grained, and mechanically excellent – with hardness and elastic modulus values far beyond anything the LLNL researchers encountered in their “SiC”.
The Legacy Problem
Unfortunately, the LLNL’s results had already been published and widely circulated. For years afterward, armor designers looked at that 0.76 figure of merit and concluded that SiC wasn’t worth serious consideration. Why bother with a material that barely outperformed cheap alumina and fell far short of B4C?
It took considerable time for the armor community to recognize that Prochazka’s sintered SiC was, for all practical purposes, a different material entirely. The chemistry was nominally the same – silicon carbide is silicon carbide – but the microstructure, density, and mechanical properties were transformed.
Today, SiC is one of the dominant materials in ceramic armor. It offers an exceptional combination of high hardness, good stability, moderate density, and acceptable multi-hit capability. The best commercial grades achieve densities above 3.15 g/cm³ (theoretical density is 3.21 g/cm³), with Vickers hardness values exceeding 2500 HV and fracture toughness around 4–5 MPa·m^½.
The LLNL’s poor results weren’t wrong, exactly – they accurately reported what they observed. But what they observed was a material that no longer exists in any meaningful sense. Their “SiC” and modern sintered SiC share a name and a chemical formula, but little else.

